Article Review: Vancomycin based tracers guiding in situ visualization of bacteria on osteosynthesis devices and surgical debridement
Objectives
- Presents the combined first-time application of a bacteria-targeted positron emission tomography (PET) tracer ([18F]PQ-VE1-vancomycin) and a near-infrared fluorescent tracer (vancomycin-IRDye800CW).
- Demonstrates the use of these tracers for non-invasive detection (PET) and real-time, fluorescence-guided surgical debridement (Fluorescence Imaging - FLI) of Gram-positive bacterial biofilms on osteosynthesis implants in a human post-mortem model.
- Shows potential for improved diagnosis and management of fracture-related infections (FRI) by distinguishing infected areas and assessing debridement effectiveness.
Methodology
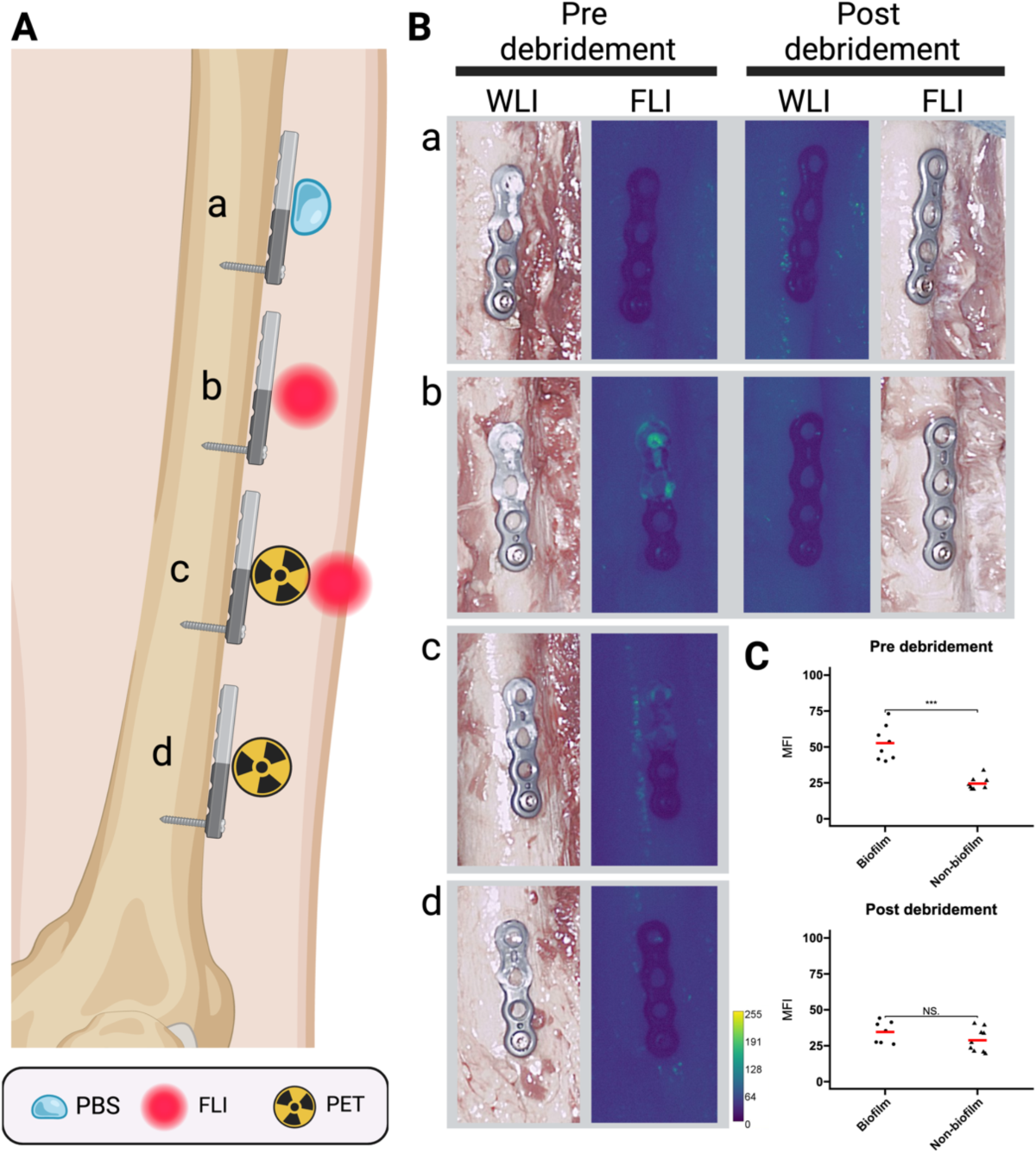
- Study utilized a Thiel-embalmed human cadaver specimen.
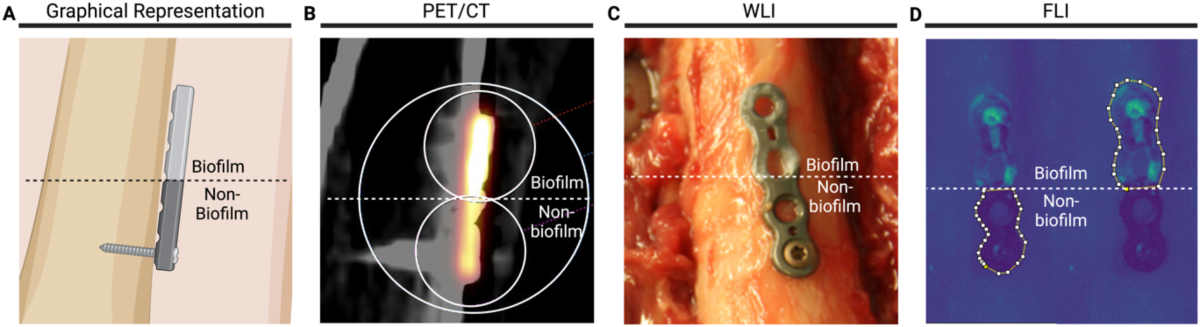
- Titanium osteosynthesis plates were partially covered with Staphylococcus epidermidis biofilm and incubated with tracers ([18F]PQ-VE1-vancomycin, vancomycin-IRDye800CW, or both) or phosphate-buffered saline (PBS).
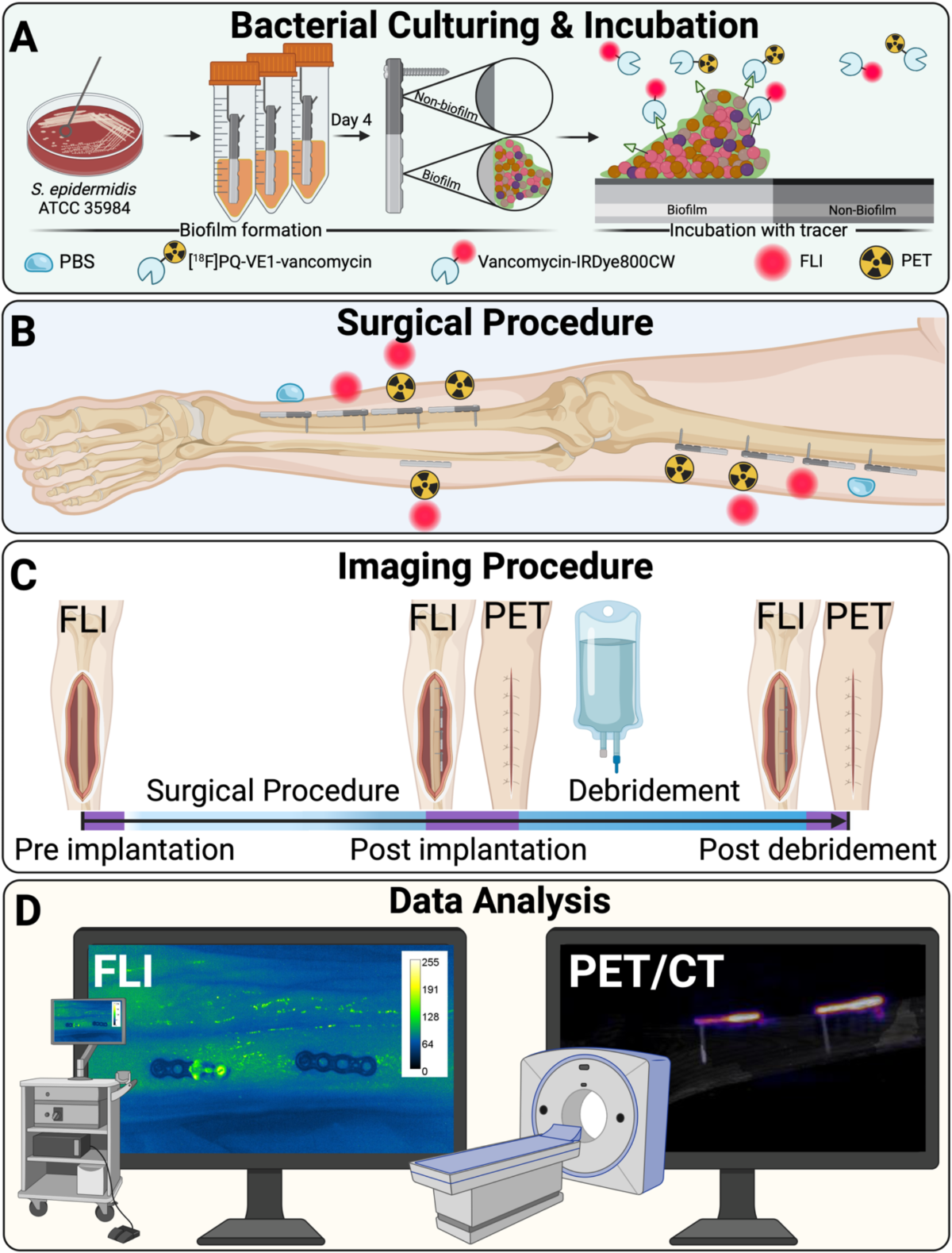
- Plates were fixed to the tibia and femur of the cadaver legs.
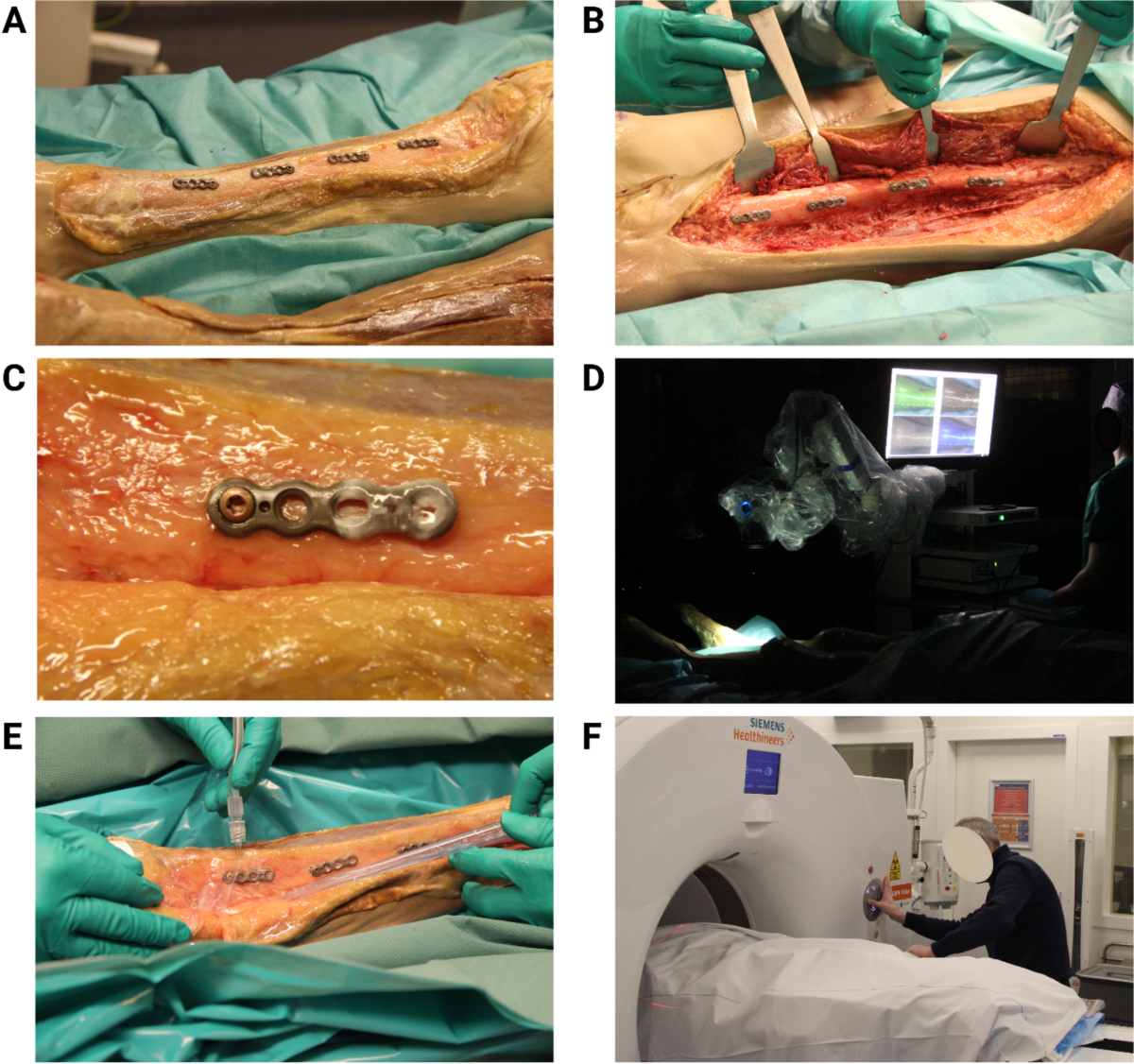
- PET combined with computed tomography (PET/CT) and FLI scans were performed before and after surgical debridement (using 3L saline and gauze).
- PET data quantification: Volumes of Interest (VOIs) were drawn on PET images using Syngo.via software to calculate mean activity (Bq/mL).
- FLI data quantification: Regions of Interest (ROIs) were drawn on fluorescence images using ImageJ to calculate mean fluorescence intensity (MFI, arbitrary units).
- Statistical analysis: Ratios between biofilm-covered and sterile sides, and pre- vs. post-debridement signals were calculated. Non-parametric Mann-Whitney U test determined statistical significance (p<0.05).
Results
- PET Imaging: Pre-debridement, [18F]PQ-VE1-vancomycin uptake was significantly higher (2.2 ± 0.6 fold) on the biofilm-covered side compared to the non-biofilm side (p<0.001). Post-debridement, the PET signal was marginal (ratio reduced to 0.9 ± 0.3), indicating effective biofilm removal (biofilm side pre- vs. post-debridement ratio: 3.6 ± 0.9).
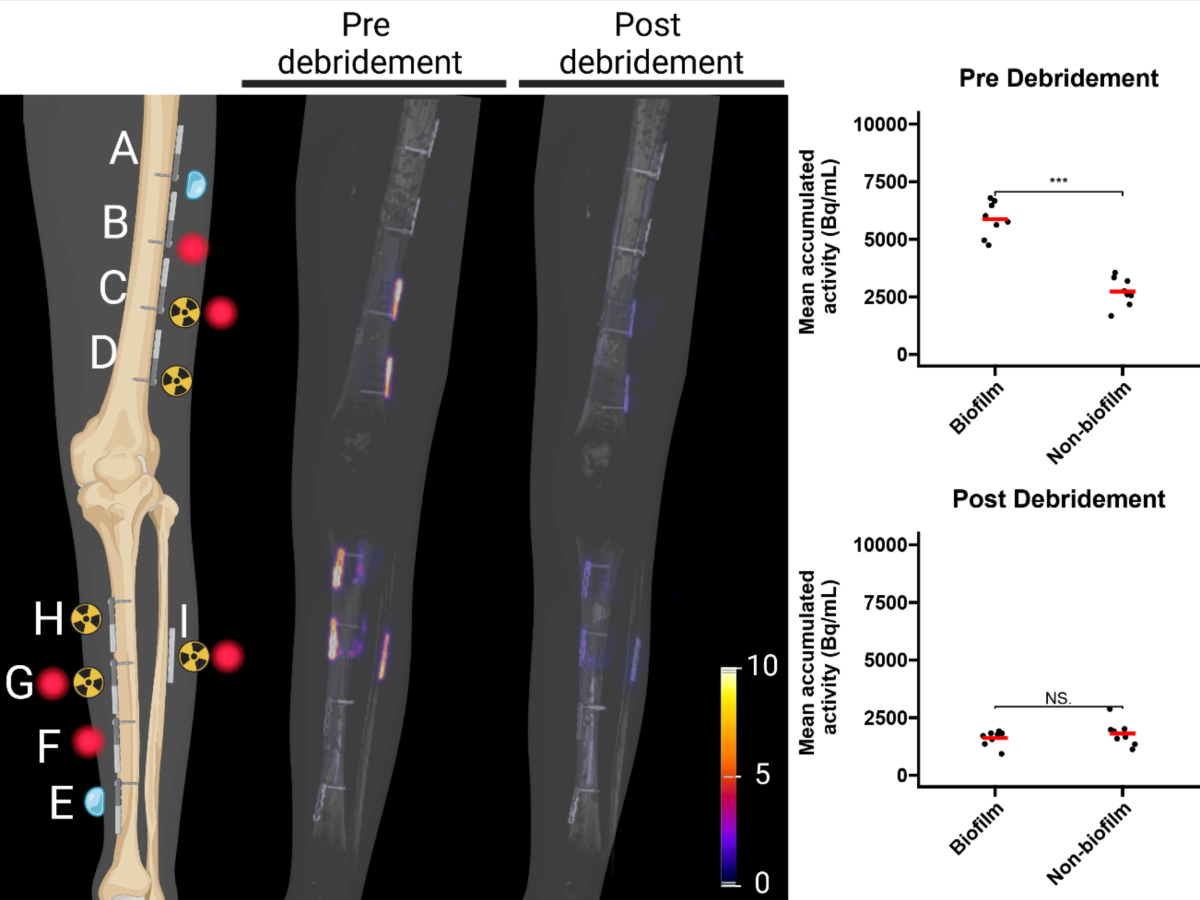
- FLI: Pre-debridement, vancomycin-IRDye800CW signal was significantly higher (2.1 ± 0.6 fold) on the biofilm-covered side compared to the non-biofilm side. Post-debridement, the signal was significantly reduced (ratio reduced to 1.2 ± 0.4), indicating removal from exposed surfaces (biofilm side pre- vs. post-debridement ratio: 1.5 ± 0.5, p<0.001).
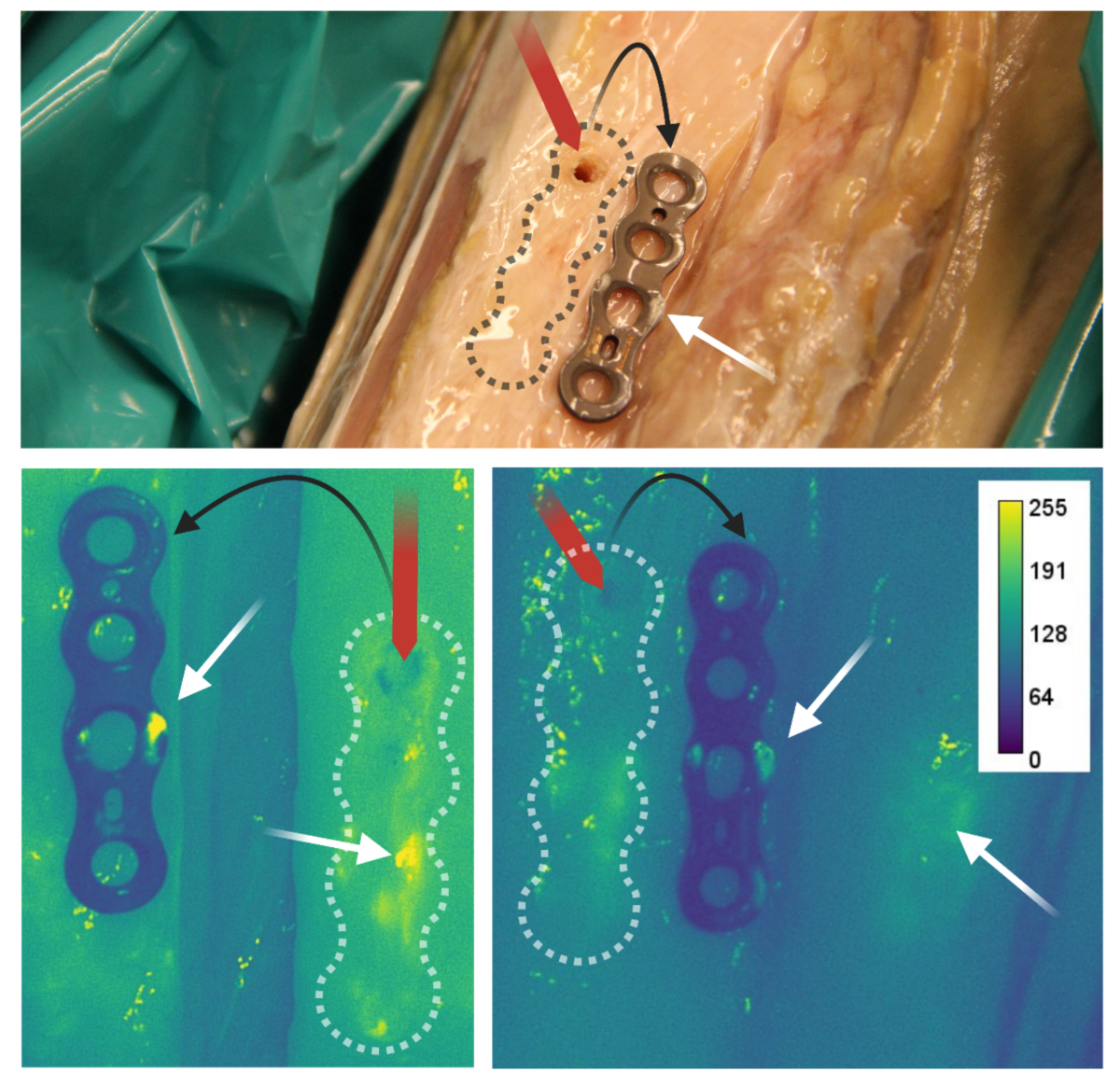
- FLI revealed residual biofilm on the undersurface of removed plates and the corresponding bone interface, which was not visible in situ.
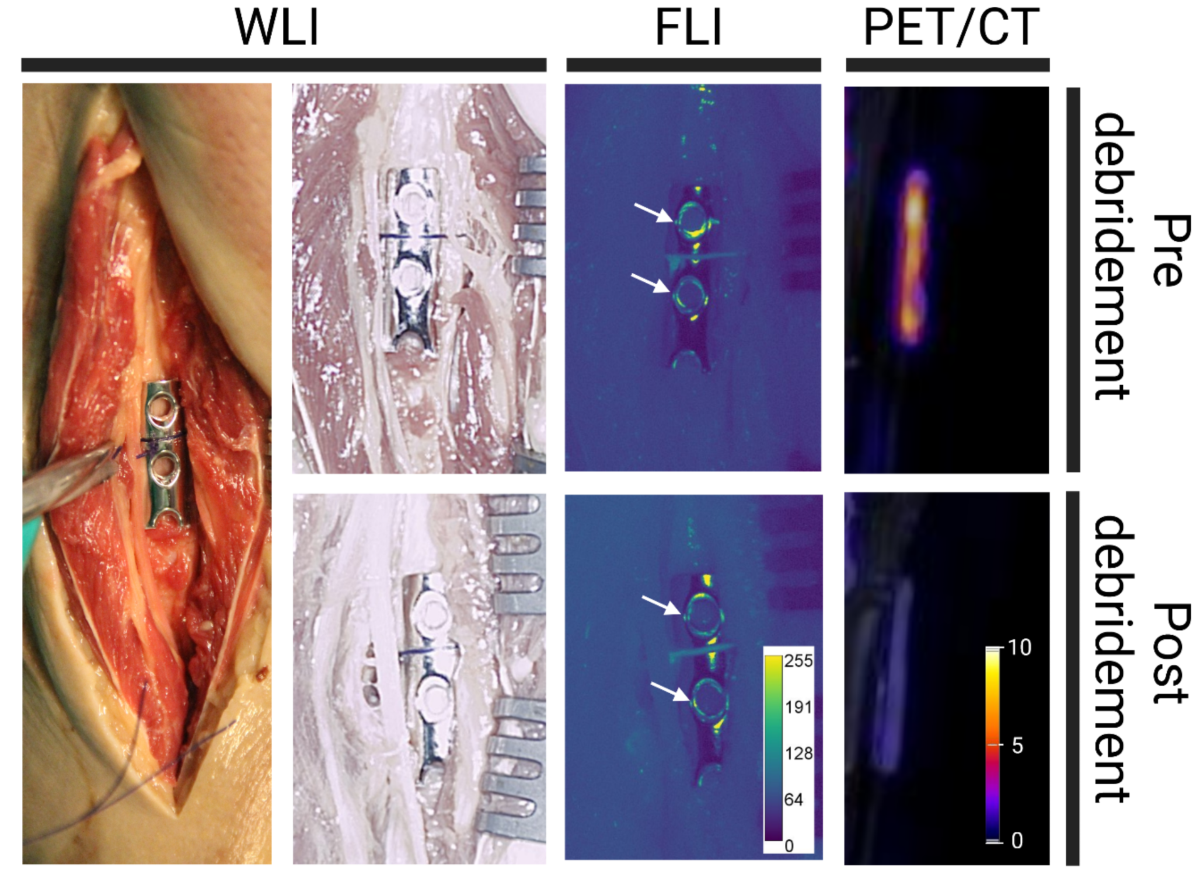
- Similar detection patterns were observed using PET/CT and FLI on osteosynthesis plates extracted from a patient with a confirmed Staphylococcus aureus infection.
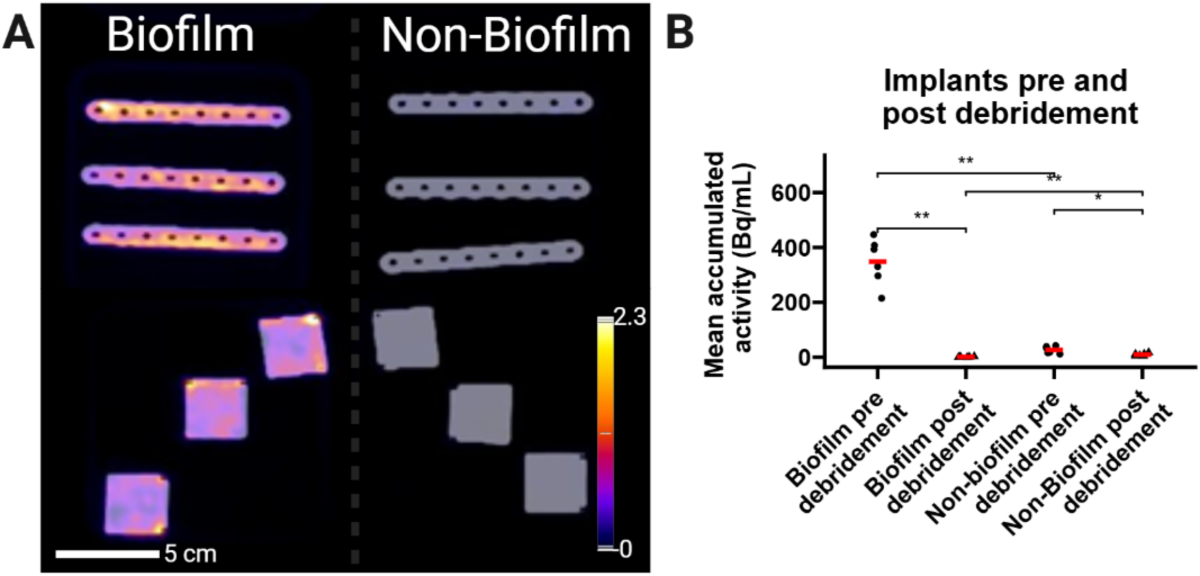
- Additional in vitro experiments confirmed higher [18F]PQ-VE1-vancomycin activity on fully biofilm-coated plates compared to sterile controls.
Discussions
- Limitation: The study utilizes a post-mortem human model, which lacks physiological processes like blood flow and immune response. This significantly differs from the clinical scenario and impacts tracer delivery dynamics, as tracers were pre-incubated rather than systemically administered.
- Limitation: Pre-incubation of tracers directly onto the biofilm bypasses the critical challenge of in vivo tracer delivery, penetration into the biofilm matrix, and clearance from surrounding tissues.
- Limitation: While FLI demonstrated residual biofilm on the implant undersurface post-removal , the study does not quantify this residual load or discuss the implications for clinical debridement success, especially when implants are retained.
- Suggestion: Future research should prioritize evaluating tracer pharmacokinetics (delivery, specific binding kinetics, and clearance rates) in a relevant in vivo animal model of FRI before proceeding to clinical trials.
- Suggestion: Determine the minimum detectable biofilm burden for both [18F]PQ-VE1-vancomycin PET and vancomycin-IRDye800CW FLI under more realistic in vivo conditions, considering potential background signal from inflammation or non-specific binding.
- Suggestion: Investigate methods to enhance the detection of biofilms on the implant undersurface and at the bone-implant interface in situ, potentially through alternative imaging approaches or tracer designs.