Article Review: Chinese management guidelines for radioactive iodine refractory differentiated thyroid cancer 2025 edition
Objectives
- Updated guidelines for the diagnosis and treatment of radioactive iodine-refractory differentiated thyroid cancer (RAIR-DTC) in China.
- Optimized definition criteria for RAIR-DTC.
- Emphasized comprehensive pre-treatment clinical evaluation and tailored individualized decision-making.
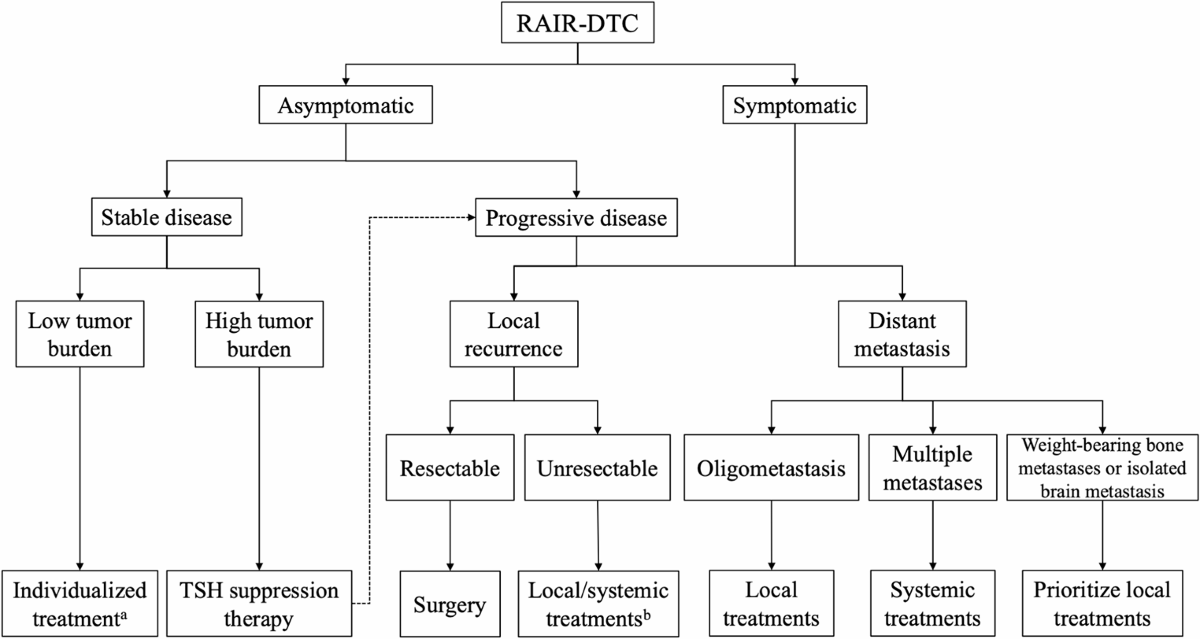
- Reviewed and updated recommendations on systematic therapy, surgery, and redifferentiation followed by RAI therapy.
- Highlighted the role of molecular imaging in pre-assessment and therapeutic response evaluation.
Methodology
- Utilized the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) approach for quantitative and qualitative evaluation of the evidence.
- Referenced prior guidelines, expert consensus, and systematic reviews.
- Addressed 26 clinical questions and formed 35 recommendations based on the evidence.
Results
- The guideline optimized the definition of RAIR-DTC, stating disease progression occurs within 1 year after RAI therapy (RAIT).
- Molecular testing is recommended, especially for DHGTC and PDTC, which account for approximately 50% of RAIR-DTCs.
- Insignificant decrease of Tg after RAIT may indicate the possibility of RAIR.
- 18F-FDG PET/CT is effective for assessing glucose metabolism in non-RAI-avid DTC lesions.
- For patients with a high tumor burden (single lesion diameter >1–2 cm or the sum of diameter of target lesions ≥4 cm), individualized management is suggested.
- Active treatment is suggested for patients with disease progression within 1 year, while regular (every 3–12 months) follow-up can be adopted in those with stable disease.
- Multi-targeted TKIs are recommended as the first-line treatment, with specific RET or TRK inhibitors for patients with corresponding gene fusions.
- RECIST 1.1 is recommended to evaluate therapeutic efficacy.
Discussions
- The guidelines provide a comprehensive overview of RAIR-DTC management, but the strength of recommendations varies significantly (from strong to weak, and high to low-quality evidence). This reflects the inherent challenges in RAIR-DTC research due to disease heterogeneity and limited high-quality evidence in some areas.
- While the guidelines emphasize individualized decision-making, more specific guidance on how to integrate various clinical factors (tumor burden, progression rate, etc.) into a concrete treatment algorithm could be beneficial.
- The guidelines mention several emerging molecular imaging techniques (RGD PET, PSMA-targeted PET, FAP inhibitors), but their clinical utility and availability in China are not fully clarified. Further research is needed to establish their role in routine clinical practice.
- The section on systemic therapies provides a good overview of available options, but the discussion on second-line treatments after TKI failure is limited. More specific recommendations based on available evidence, even if limited, would be helpful.
- The guidelines acknowledge the limited efficacy of immunotherapy in most RAIR-DTC cases, which is consistent with current evidence. However, the potential role of combination therapies (e.g., lenvatinib plus pembrolizumab) could be discussed in more detail, including the need for further research to confirm their superiority over monotherapy.